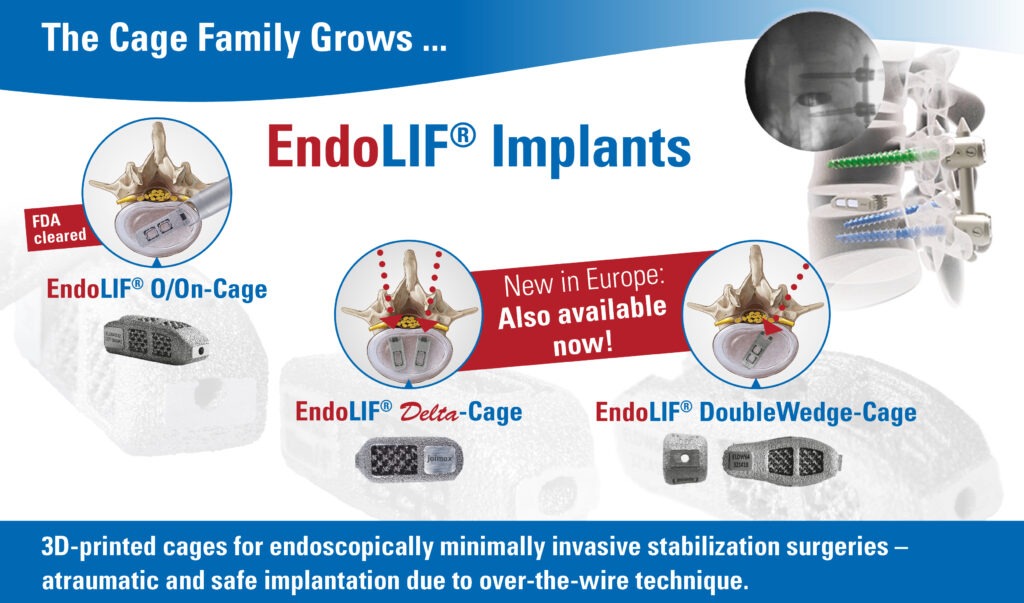
Irvine, California – July 13, 2015 (BUSINESS WIRE) – The German company joimax®, developer of technologies and training methods for minimally invasive endoscopic spinal surgery, today announced it received 510(k) clearance from the U.S. Food and Drug Administration (FDA) to market its Endoscopic Lumbar Interbody Fusion, or EndoLIF® On-Cage.
The EndoLIF On-Cage® consists of titanium alloy, produced with Electron Beam Melt (EBM) technology. The cage displays a porous surface with diamond cell structure, providing an optimal base for cell proliferation and bone growth. Two large openings, which may be filled with autogenous bone, support the creation of a straight column for fusion.
The EndoLIF® implant allows surgeons to utilize an inter-muscular approach, similar to a mini transforaminal lumbar interbody fusion (TLIF), into the intervertebral disc, enabling endoscopic-assisted fusion. Dr. Ralf Wagner, LIGAMENTA Spine Center, Frankfurt and Dr. Bernd Illerhaus, ONZ, Datteln/Recklinghausen, two German spine specialists, have already performed more than 200 out of 600 EndoLIF procedures in Europe. “The access is dura and nerve-gentle, preserves the dorsal bony structures and we can avoid scar tissue because of the stepwise tissue dilation,” said Dr. Illerhaus.
The EndoLIF® On-Cage is designed to be used with supplemental posterior fixation, such as the joimax Percusys® percutaneous pedicle screw-rod system. Cage implantation can be performed with a posterior or postero-lateral approach, either using an open or endoscopic-assisted method.
“With the EndoLIF® program, joimax offers a complete endoscopic-assisted solution for spinal stabilization and fusion. In the future, we will be able to treat patients with even more gentle techniques,” comments Wolfgang Ries, CEO and founder of joimax®. “Our next development will be an EndoLIF® Cage on the basis of our iLESSYS® Delta system for posterior lumbar inter-body fusion (PLIF).”