Karlsruhe, GER– March 12, 2019 – Earlier this month, at the international training summit dubbed, Faculty and User Meeting, joimax®, the Germany-based market leader of technologies and training methods for full-endoscopic minimally-invasive spinal surgery, presented its newest products and technologies. During the two-day event, joimax® also honored four surgeons with the Kambin Award and introduced the Endoscopic Spine Academy, ESPINEA® .
The summit took place at the Paracelsus Medical University (PMU) in Salzburg, Austria, March 1-2, 2019 where more than 100 participants attended top-class presentations as well as well a human cadaver lab. The lab was equipped with ten hands-on training stations: six cadaveric stations with 4K image resolution equipment and within those stations, two Intracs® navigation systems where integrated; two stations with Real Spine simulation models; and two stations with the latest EndoTrainer® Plus virtual simulator, which can be used in any lab for training.
The Intracs® em electromagnetic navigation system for endoscopic minimally invasive spine surgery in lumbar and thoracic applications is very easy to set up, extremely user-friendly, and ready to serve as a stand-alone device. Specifically designed for simple and safe access to any spinal target, the system relies on electromagnetic tracking and allows for simultaneous navigation of multiple instruments such as needles, guiding rods, reamers, and endoscopes, also developed by joimax®.
Dr. Erik Traupe (Germering, GER) stated: “I am really surprised how precise and easy navigation can be.”
After multiple successful introductions in 2018, Intracs® em is available globally, except in the United States, where FDA submission is planned in spring 2019.
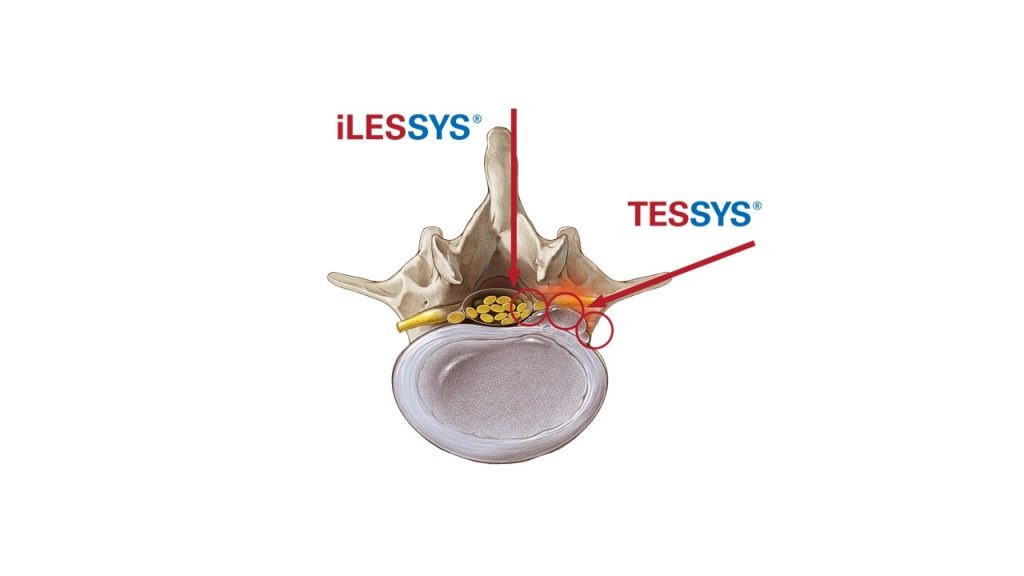
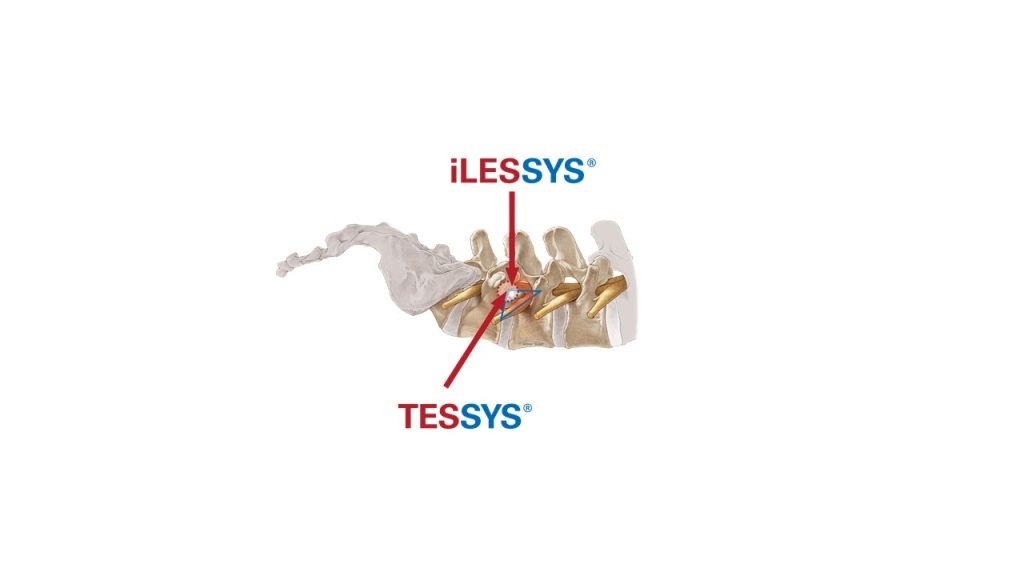
Another trendsetting innovation presented at the summit: the joimax® TESSYS® Thx, a derivative of the well-known TESSYS®. Designed for a comfortable and safe treatment of decompressions in the thoracic region, it combines a special endoscope for thoracic applications, a viewing angle with 45° adapted to a steeper access route, a reduced diameter and a big working channel.
Jun Seok Bae, MD of the Wooridul Spine Hospital Group in South Korea is very excited about the advantages: “… just great, I want it a.s.a.p. and am ready in my hospital for daily usage!”
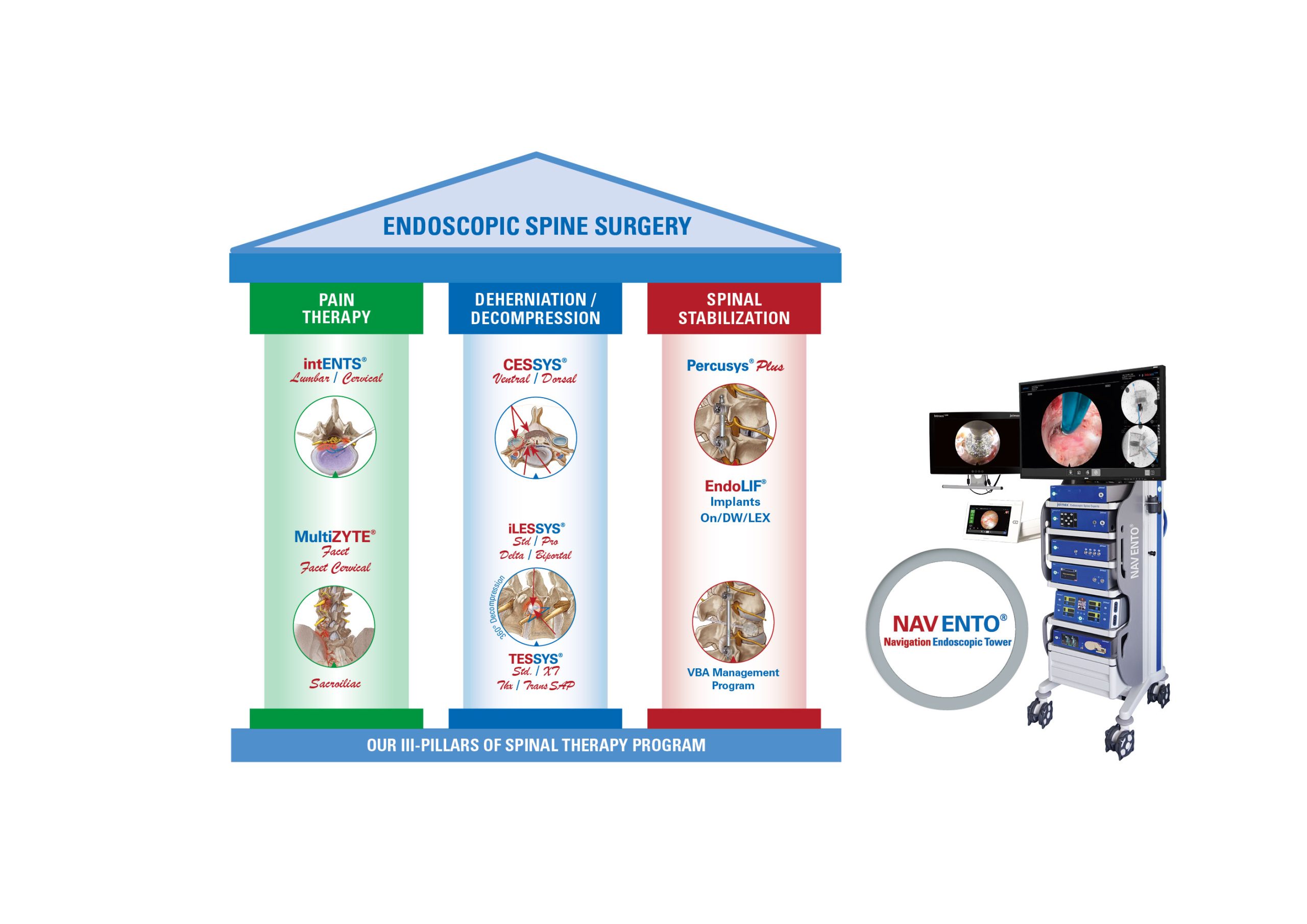
Moreover, joimax® showcased its EndoLIF® product line: the well-known 3D-printed EndoLIF® O/On-Cage, the EndoLIF® Delta Cage, and the latest EndoLIF® Double Wedge Cage, where approval is expected shortly.
Both Dr. Ralf Wagner (Frankfurt, GER) and Dr. Paul Houle (Cape Cod, MA, USA) are convinced of this solution for spinal fusion: “With the gentle and atraumatic access via gradual tissue dilatation, muscles remain intact and infection risk is reduced. Using 3D-printing technology, an open diamond cell structure, and a rough and porous surface, the implant ensures optimal bone ingrowth, stability and finally fusion.”
During the summit, the Parviz Kambin-Award was presented under the patronage of the PMU, ESPINEA and joimax® in recognition and honor of outstanding scientific and educational dedication to transforaminal surgery to Christoph P. Hofstetter (Seattle, WA, USA), Jian Shen (Amsterdam, NY, USA), Albert Telfeian (Providence, RI, USA) and Dinh Ngoc Son (Hanoi, Vietnam).
“I am thrilled about the Kambin Award, the most prestigious award in endoscopic spine surgery,” said Jian Shen.