Karlsruhe – joimax® is participating with an industrial exhibition at the annual meeting of the european spine society „EUROSPINE“ from October 1st to October 3rd at the Congress Center in Lyon. In Forum N° A43 the company, located in Karlsruhe, exhibits its now highly diversified product portfolio for 360° surgery in nearly all areas of the spine including endoscopic or percutaneous stabilization. In various lunch symposia, numerous european experts will show their experiences and results. During a 2 hours lunch symposium on the first day of the Congress in Hall Rhône 1 internationally recognized spine specialists under the leadership of Dr. Menno Iprenburg (Veenhuizen / Netherlands) and Dr. Alastair Gibson (Edinburgh / United Kingdom) will provide insights of their experience and into their scientific results.
Lectures on the latest product developments are expected with great interest, hereby:
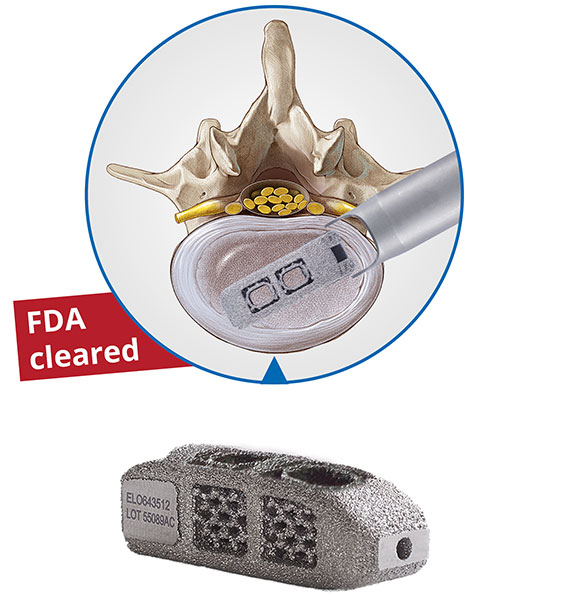
- The EndoLIF® system for endoscopic lumbar column stabilization. This system includes not only the fully endoscopic access technology to the disc space with the proven TESSYS® or iLESSYS® method, but also an implant, the so-called O-Cage (O = oblique).
- Percusys®, an innovative screw and rod system for the posterior percutaneous stabilization.

The advantages of this methods are obvious:
- Tissue sparing access by dilation
- Protection of dura and nerves through a proven access corridor
- Preservation of the posterior bony structures due to the special lateral access
- EndoLIF® O-Cage made of Ti6Al4V for optimized bone ingrowth
- Percutaneous stabilization by Percusys® and therefore minimally invasive
Both new systems are CE approved and the FDA approval is in progress.
Since 2004, more than 100,000 patients in 30 countries were successfully operated using the TESSYS® method and their further developments. Studies show the shorter reconvalescence time, faster recovery and thereby speed up the return of the patients to work and enhance quality of life.
Visit joimax® at EUROSPINE, Cité Internationale, Lyon Convention Centre, Forum N° A 43 and see for yourself the benefits of the new systems.